Why Medications Cause Different Side Effects in Different People
CYP2D6 Metabolizer Status Calculator
Understand Your CYP2D6 Metabolizer Status
Genetic variations in your CYP2D6 enzyme can dramatically affect how your body processes many medications. This tool shows how your metabolizer status impacts drug effectiveness and side effects.
Based on your selection, you'll see what this means for common medications and what you should discuss with your healthcare provider.
Results
Key Information
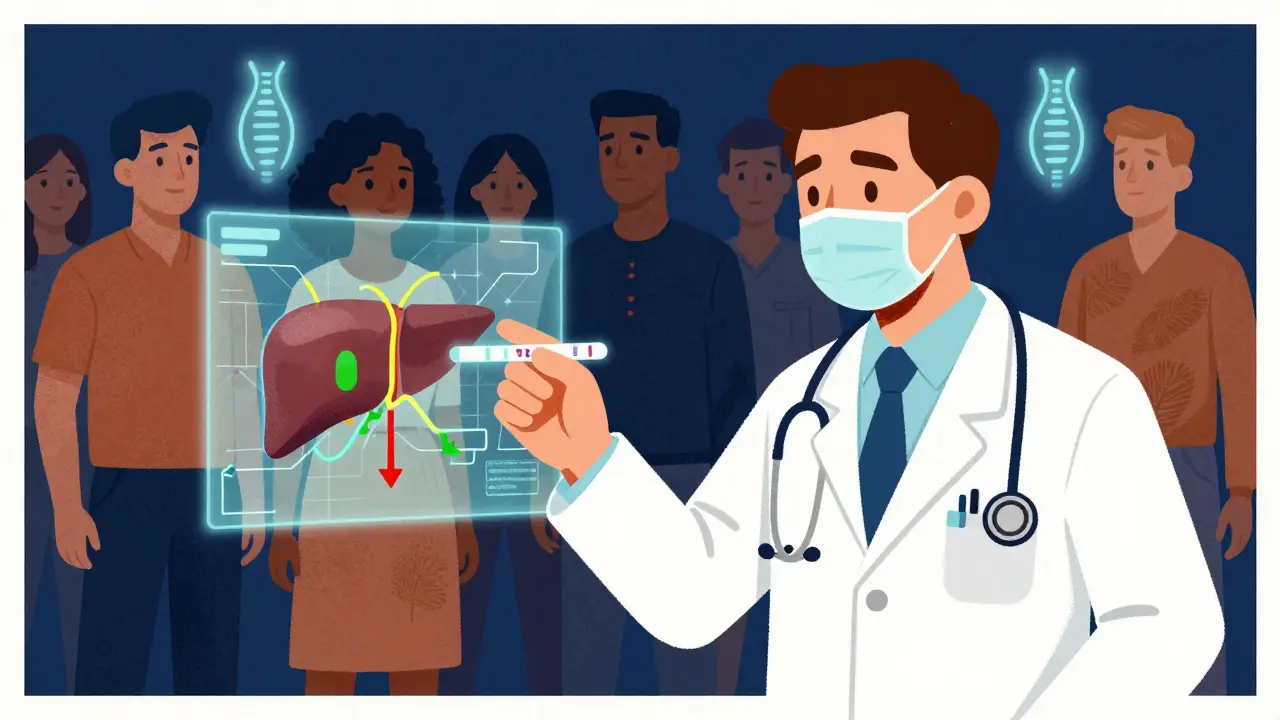
CYP2D6 is a liver enzyme that processes about 25% of all medications, including many antidepressants, painkillers, and heart medications.
Understanding your metabolizer status can help prevent dangerous side effects and ensure medications work effectively.
Genetic testing for CYP2D6 is now covered by many insurers and costs as little as $250.
Have you ever heard someone say, "I took that same pill and felt fine," while you ended up dizzy, nauseous, or worse? It’s not just you. Medications don’t work the same way for everyone. One person might need a single dose of a drug to feel better, while another takes the same dose and ends up in the hospital. This isn’t luck. It’s biology. And understanding why this happens could save lives.
Genes Are the Main Player
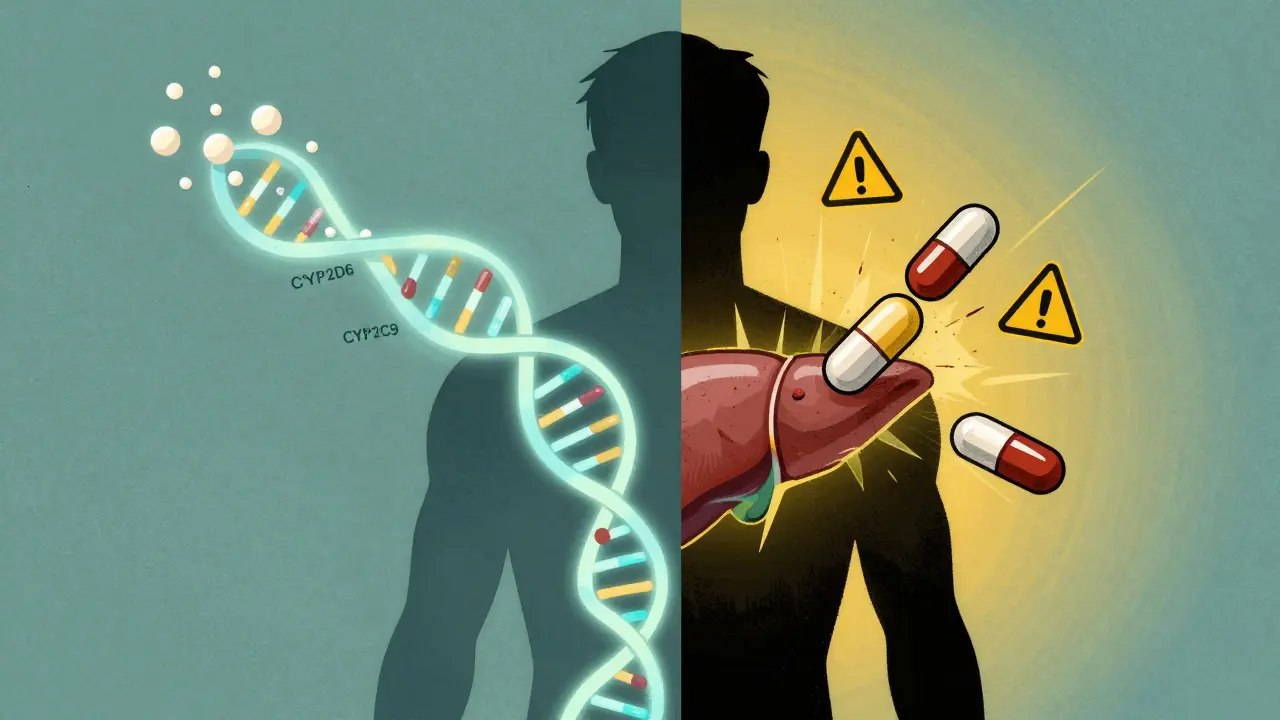
Your genes aren’t just about eye color or height. They’re also the blueprint for how your body handles drugs. Around 20% to 95% of how you respond to a medication comes down to your DNA - yes, that much. The biggest players? Enzymes in your liver, especially the cytochrome P450 family. These enzymes break down drugs so your body can get rid of them. But not everyone has the same version of these enzymes.
Take CYP2D6. About 5-10% of white Europeans are "poor metabolizers" - their bodies barely break down certain drugs. That means the drug builds up, and side effects skyrocket. On the flip side, 1-2% of Europeans, and up to 29% of Ethiopians, are "ultra-rapid metabolizers." Their bodies clear the drug so fast it never works. A painkiller might vanish from their system in an hour. They end up taking more, thinking it’s not working - and accidentally overdose.
This isn’t theoretical. Warfarin, a blood thinner, is a classic example. Two genes - CYP2C9 and VKORC1 - explain 30-50% of why people need totally different doses. One person might need 5 mg a day. Another, with different genes, needs 1 mg. Get it wrong, and you risk a stroke or a bleed. Studies show that when doctors test for these genes before prescribing warfarin, patients reach safe levels 27% faster and have 31% fewer major bleeds.
Age, Body Fat, and Other Physical Changes
Genes aren’t the whole story. Your body changes as you age. Older adults carry more body fat and less water. That means fat-soluble drugs - like some antidepressants or sleep aids - get stored in fat tissue and hang around much longer. One study found elderly patients have 30-40% more fat storage than young adults. So a dose that was fine at 40 might be dangerous at 70.
Also, kidney and liver function decline with age. These organs clear drugs from your system. When they slow down, drugs stick around. That’s why older patients are 300% more likely to have serious side effects when taking five or more medications. It’s not just the drugs - it’s the pile-up.
What You Eat, Drink, and Take Alongside
Drugs don’t live in a vacuum. Grapefruit juice? It blocks a liver enzyme (CYP3A4) that breaks down dozens of medications. One glass can double or triple the drug’s effect. That’s why some statins and blood pressure pills come with warning labels.
Then there’s inflammation. If you’ve got an infection, arthritis, or even a bad cold, your body releases chemicals that can cut liver enzyme activity by 20-50%. Suddenly, a drug you’ve taken safely for years starts building up. A 2021 study showed that even mild inflammation can turn a safe dose into a toxic one.
And don’t forget drug interactions. Amiodarone, a heart medication, can block warfarin’s breakdown. The result? Warfarin levels jump 100-300%. That’s not a typo. It’s a bleeding emergency waiting to happen. This is why pharmacists check your full list of meds - not just the new one.

Genetics and Real-World Outcomes
The data doesn’t lie. A 2022 Mayo Clinic study followed 10,000 patients. Those who got genetic testing before being prescribed drugs had 32% fewer emergency room visits and 26% shorter hospital stays. That’s not just numbers - it’s fewer scary nights in the ER, less time away from work, less stress for families.
In asthma, a genetic variant in the LTC4 synthase gene affects 15% of severe cases. Those patients respond dramatically better to leukotriene modifiers like zafirlukast - 45% better lung function improvement. But if you don’t have that variant? You get the same expensive pill, pay $250-$300 a month, and feel nothing. It’s waste. And it’s common.
One JAMA Internal Medicine case described a 68-year-old woman who kept having dangerous bleeding episodes. Her INR kept climbing above 10 - a life-threatening level. Doctors adjusted her warfarin dose again and again. Nothing worked. Then, genetic testing revealed she had two copies of the CYP2C9*3 variant - a rare "poor metabolizer" status. Her dose was cut by 60%. Within weeks, she was stable. No more bleeding. Just one test changed everything.
Why We’re Not Doing This More Often
So if it works, why isn’t everyone getting tested?
The FDA has added pharmacogenomic info to over 300 drug labels. For 44 of them, they give specific dosing advice based on genes. But only 18% of U.S. insurers cover testing. Most doctors haven’t been trained to interpret the reports. A 2023 survey found 68% of physicians feel unprepared to use genetic data. And that’s after 30 years of research.
There’s also a gap in coverage. Testing just CYP2C9, CYP2C19, and CYP2D6 explains only 15-19% of all adverse reactions. But hundreds - maybe thousands - of other genes are involved. A person might have a "perfect" profile for those three enzymes and still react badly because of a gene nobody tests for.
And then there’s polypharmacy. An elderly patient on six medications? The interactions become a maze. Gene A affects drug 1. Drug 2 blocks the enzyme for drug 3. Drug 4 is cleared by the same liver pathway as drug 5. It’s not just genetics - it’s a perfect storm.
What’s Changing Now
The tide is turning. In January 2024, Medicare started covering pharmacogenomic testing for 17 high-risk drugs. The first point-of-care CYP2C19 test - which gives results in 60 minutes - was approved by the FDA in 2023. That’s huge. Now, a cardiologist can test for clopidogrel response right in the office before handing out a prescription.
Europe just mandated that all new clinical trials include pharmacogenomic analysis. In the U.S., the NIH’s PGRN database now includes genetic data from 1.2 million patients across 15 healthcare systems. Oncology leads the charge - 65% of cancer centers use this routinely. Psychiatry and cardiology are catching up. Primary care? Still behind.
And costs? They’ve dropped from $2,000 per gene panel in 2015 to around $250 today. That’s within reach for most clinics. The real barrier isn’t money anymore - it’s systems. Most electronic health records don’t alert doctors when a patient’s genes conflict with a prescribed drug. Imagine if your phone warned you before you clicked a dangerous link. That’s what we need.
What You Can Do
If you’re on multiple medications - especially blood thinners, antidepressants, painkillers, or heart drugs - ask your doctor: "Has my genetic profile been considered?" You don’t need a full genome scan. Just testing for a few key enzymes can make a big difference.
Keep a list of all your meds - including supplements and OTC drugs. Bring it to every appointment. Tell your doctor about any strange side effects, even if they seemed minor. That "mild nausea" after starting a new pill? It might be a red flag.
And if you’ve had a bad reaction before, don’t assume it’s "just you." You might have a genetic variation that’s common, but rarely tested. Your experience matters. It’s not anecdotal - it’s data waiting to be understood.
Medications aren’t one-size-fits-all. They never were. The future of medicine isn’t just better drugs - it’s smarter prescribing. And it starts with knowing who you are - down to your genes.
Why do some people have side effects while others don’t, even with the same drug?
It’s mostly due to genetic differences, especially in liver enzymes like CYP2D6 and CYP2C19 that break down drugs. Some people metabolize drugs too slowly, causing buildup and toxicity. Others process them too fast, making the drug ineffective. Age, body composition, other medications, and even inflammation can also change how a drug behaves in the body.
Is genetic testing for drug reactions worth it?
For certain high-risk drugs like warfarin, clopidogrel, and some antidepressants, yes. Studies show it reduces hospitalizations by up to 31% and cuts emergency visits by 32%. If you’re on multiple medications, have had bad reactions before, or are over 65, the benefits often outweigh the cost - especially since testing now costs as little as $250.
Can I get tested without a doctor’s order?
Some direct-to-consumer tests offer pharmacogenetic info, but they’re not always clinically validated. The most reliable results come through a healthcare provider who can interpret the data in context with your health history and current medications. A pharmacist or genetic counselor can help you understand what the results mean for your treatment.
Do I need to get tested every time I start a new drug?
No. Your genes don’t change. Once you’ve had a pharmacogenomic test, the results apply to any drug that’s affected by those genes. The key is keeping your results on file and sharing them with every provider who prescribes you medication. Think of it like a blood type - it’s lifelong information.
Are there drugs that don’t respond to genetic testing?
Yes. For some medications, especially those used in complex conditions like cancer or mental health, dozens of genes play a role - not just one or two. Right now, genetic testing works best for drugs with clear, single-gene links - like warfarin or clopidogrel. As polygenic risk scores improve, we’ll be able to predict responses for more drugs, but for now, it’s not universal.




10 Comments
jared baker
March 18 2026I've seen this firsthand. My grandma was on warfarin for years, and they kept adjusting her dose because her INR kept spiking. No one thought to test her genes until she ended up in the ER. Turns out she had CYP2C9*3. Once they cut her dose by 60%, she stopped bleeding and could finally sleep through the night. Simple test. Life-changing result.
Michelle Jackson
March 20 2026People act like this is some revolutionary breakthrough but it's just biology. We've known for decades that bodies handle drugs differently. The real problem? Doctors who refuse to listen to patients and rely on cookie-cutter protocols. If you're still prescribing the same dose to a 70-year-old as you would to a 30-year-old, you're not a doctor-you're a liability.
Suchi G.
March 22 2026I live in India, and here, most people don't even know what a pharmacist is, let alone pharmacogenomics. My uncle took the same blood thinner as his brother and ended up in intensive care. The doctors blamed his diet, his stress, his age-but never checked his genes. I cried when I read this article because it's not just about science-it's about dignity. People deserve to know why their bodies betray them. It's not weakness. It's not bad luck. It's just code. And we're ignoring the manual.
Andrew Muchmore
March 23 2026Test your genes once. Keep the results. Share them with every provider. That's it. No more guessing. No more trial and error. Just facts.
Paul Ratliff
March 23 2026grapefruit juice and warfarin = bad combo. learned that the hard way. my buddy took his meds with a glass of juice and ended up in the hospital. doc said 'i didn't even know that was a thing' lol. we need better education.
SNEHA GUPTA
March 23 2026It’s strange how we accept that every car has a manual but we treat human biology like it’s a black box. We don’t just ‘take a pill’-we are complex systems of enzymes, receptors, transporters, and metabolic pathways. Yet we expect a one-size-fits-all approach. The irony? The same people who demand personalized nutrition, fitness, and skincare refuse to consider that their genes might dictate how their body processes medicine. This isn’t sci-fi. It’s the next logical step in medicine. And we’re still arguing about whether it’s worth it.
David Robinson
March 25 2026They say testing helps but let’s be real-most docs don’t know how to read the reports. I work in a clinic. We got the results back for a patient last month. The pharmacist flagged a dangerous interaction. The doctor looked at it for 30 seconds and said 'I'll just lower the dose.' No. You don't just 'lower' when you have a CYP2D6 poor metabolizer. You switch drugs. Or you wait. Or you consult. But you don't guess.
Jeremy Van Veelen
March 26 2026This is the future of medicine. The kind of stuff they show in those slick pharmaceutical ads where a glowing DNA strand zips through a vein and whispers, 'I know what you need.' But here’s the truth: we’re not ready. We have the science, yes. But we lack the humility. We still think we can outsmart evolution with a standard prescription pad. We’re not curing people-we’re running experiments on them. And the cost? Lives. And dignity. And trust.
Laura Gabel
March 28 2026Why are we wasting money on this? In America we got better things to spend on. My cousin took the same pill as me and was fine. Why test everyone? Just tell people to stop complaining and take their meds like normal people.
jerome Reverdy
March 29 2026As someone who's been on 8 meds for 12 years, this is the first time I felt heard. The CYP2D6 poor metabolizer thing? That's me. I thought I was just 'sensitive.' Turns out I'm a genetic outlier. My psychiatrist finally switched me to a non-metabolized alternative after seeing the report. No more dizziness. No more brain fog. I can work again. This isn't just science-it's restoration. And it's available now. If you're on polypharmacy, ask. Don't wait for a crisis. Your future self will thank you.