Carbamazepine Generics: Enzyme Induction and Drug Interactions Explained
When you take carbamazepine for epilepsy or nerve pain, you’re not just taking a pill-you’re managing a complex biological system. The drug works by calming overactive nerves, but its real challenge lies in how it changes your body’s chemistry. Carbamazepine doesn’t just sit there. It triggers your liver to produce more enzymes that break down not just itself, but also other medications you might be taking. This is called enzyme induction. And when you switch from one generic version to another, even if the label says the same thing, your body might respond differently. That’s why carbamazepine generics aren’t as interchangeable as people think.
How Carbamazepine Induces Enzymes
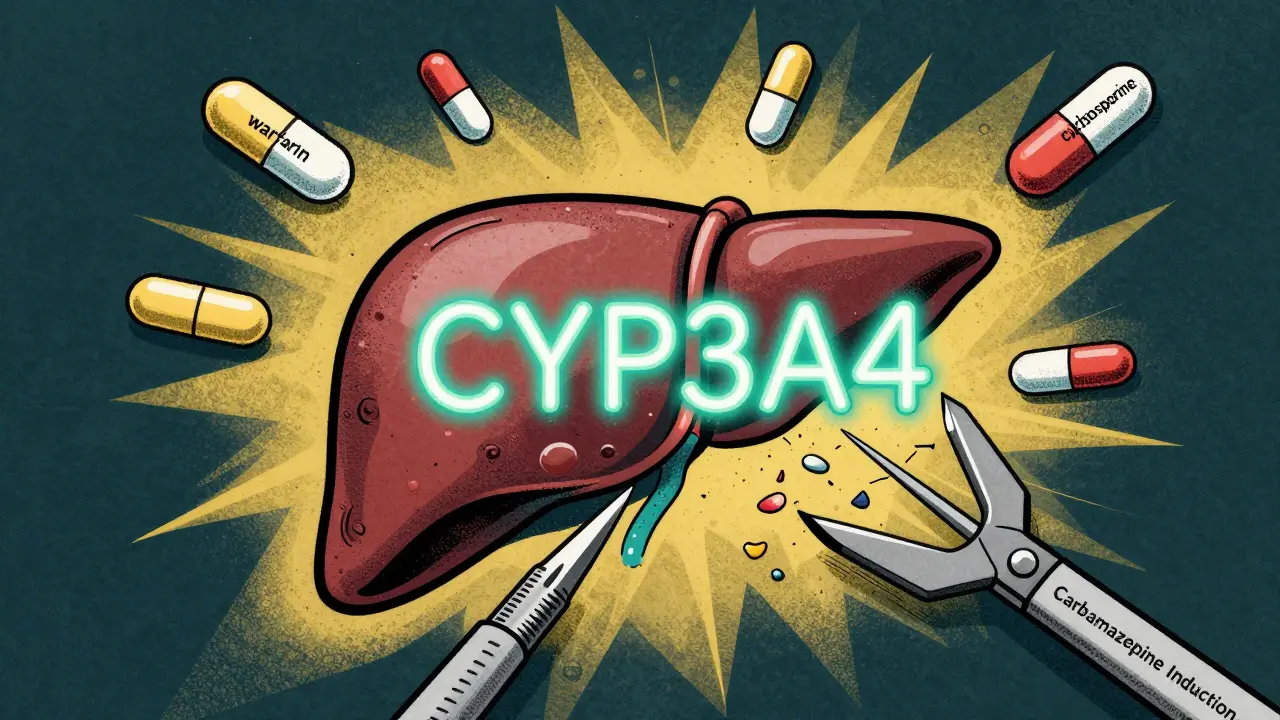
Carbamazepine is one of the strongest inducers of the CYP3A4 enzyme in the liver. This enzyme handles about half of all prescription drugs. When carbamazepine enters your system, it tells your liver to make more of this enzyme. Within 48 hours, the process starts. By two to three weeks, it’s at full force. And here’s the catch: it doesn’t just speed up its own breakdown. It speeds up the breakdown of other drugs too.
That means if you’re on warfarin for blood thinning, your INR might drop suddenly. If you’re taking cyclosporine after a transplant, your levels could crash, risking organ rejection. Even birth control pills can become less effective. The FDA lists over 20 medications with reduced effectiveness when taken with carbamazepine, including antifungals, HIV drugs, and blood pressure pills like amlodipine. The problem isn’t the dose-it’s that your body is now processing those drugs faster than before.
What makes this worse is that carbamazepine also induces UGT enzymes and P-glycoprotein, which help clear drugs from your gut and brain. So even if your liver isn’t the only issue, your body’s entire drug-clearing system is being turned up. This isn’t a minor side effect. It’s a systemic shift that can change how every other medication works in your body.
Why Generic Versions Aren’t Always the Same
All generic carbamazepine must meet FDA standards: they must deliver 80% to 125% of the brand’s absorption. That sounds precise, but it’s not enough for a drug with a narrow therapeutic window. Carbamazepine’s effective range is only 4 to 12 mcg/mL. A drop below 4 means seizures might return. A rise above 12 can cause dizziness, nausea, or even toxicity.
Studies show that when patients switch between different generic versions-even ones that are technically “bioequivalent”-about 12% experience problems. Some have more seizures. Others develop new side effects like rashes or confusion. In one study, 7.8% of those switched ended up in the emergency room. Why? Because bioequivalence is tested in healthy volunteers on a single dose. Real patients? They’re often older, on multiple drugs, or have liver or kidney issues. Their bodies handle the drug differently.
Extended-release formulations add another layer. The way the beads dissolve in your gut matters. One generic might release the drug slowly over 12 hours. Another might release it faster. For someone with gastroparesis or a slow digestive system, that difference can mean the difference between control and crisis. A nurse on Reddit noticed that one generic’s beads were larger than others, which affected how they dissolved in patients with delayed stomach emptying. That’s not something a standard bioequivalence test catches.
Gender, Genetics, and How Your Body Reacts
Men and women metabolize carbamazepine differently. Women tend to break it down faster because they have higher levels of CYP3A4. That means women often need slightly higher doses to stay in the therapeutic range. But here’s the twist: hormonal changes during the menstrual cycle, pregnancy, or while using birth control can shift that metabolism again. A 2021 study found women of childbearing age had 22% more breakthrough seizures after switching generics-likely because their enzyme activity fluctuated with hormones.
Genetics play a huge role too. About 15% of people of Asian descent carry the HLA-B*1502 gene. If they take carbamazepine, they have a 10 times higher risk of developing Stevens-Johnson Syndrome-a life-threatening skin reaction. That’s why screening is required before starting. But even beyond that, other gene variants affect how fast you process the drug. One variant, CYP3A4*22, means you need 25% less carbamazepine to reach the same blood level. Most doctors don’t test for this. But if you’ve had trouble with generics before, it might be worth asking.
Therapeutic Drug Monitoring: Your Best Defense
If you’re on carbamazepine, especially a generic version, you need regular blood tests. Not once a year. Not just when you feel off. You need them after any switch, any dose change, or if you start or stop another medication. The ideal time to test is 7 to 14 days after switching. That’s when levels stabilize.
A drop of more than 15% in your blood concentration is a red flag. It doesn’t mean the generic is bad. It means your body is handling this version differently. Your doctor might need to adjust your dose-even if you’re on the same number of milligrams. A patient on Epilepsy Foundation’s forum saw her level drop from 7.2 to 4.8 mcg/mL after switching generics. She was still on the same dose. But 4.8 is near the bottom of the safe range. No wonder her seizures increased.
Doctors who manage epilepsy know this. The American Epilepsy Society recommends keeping patients on the same manufacturer whenever possible. If you’ve been stable on one generic, don’t let your pharmacy switch you without telling you. Ask for the brand name or specific manufacturer on your prescription. Use the “dispense as written” code (DAW 1). That stops automatic substitution.
What You Can Do Right Now
- Check your current brand. Look at your pill bottle. Note the manufacturer name. If you’ve been switched before, write it down.
- Ask for a blood test. If you’ve switched generics recently, request a carbamazepine level check in 7-10 days.
- Don’t assume all generics are equal. Just because two pills say “carbamazepine 200 mg” doesn’t mean they act the same in your body.
- Tell your pharmacist. If you’ve had issues before, say so. Pharmacists can flag your profile to prevent future switches.
- Watch for new symptoms. Dizziness, rash, nausea, confusion, or more seizures? These aren’t normal. They could mean your levels dropped-or spiked.
There’s no perfect solution yet. The FDA is working on better testing methods for drugs like carbamazepine. But until then, the burden is on you and your doctor to stay vigilant. You’re not just taking a generic drug. You’re managing a delicate chemical balance. And that balance can shift with a simple pharmacy change.

What to Do If You’re Starting Carbamazepine
If you’re new to carbamazepine, start low and go slow. Your doctor should begin with a low dose and increase gradually. Monitor for side effects. Get your first blood level check at 2 weeks. If you’re of Asian descent, ask for HLA-B*1502 screening before starting. If you’re positive, carbamazepine might not be the right choice. Alternatives like levetiracetam or lacosamide don’t carry the same enzyme-inducing risks.
And if you’re on other medications? Bring your full list to every appointment. Even over-the-counter drugs like St. John’s Wort can interfere. Your pharmacist can run a drug interaction screen-but only if they know what you’re taking.
Looking Ahead: Precision Dosing
The future of carbamazepine treatment isn’t just about which generic you get. It’s about personalizing the dose based on your genes, your sex, your weight, and your other medications. Researchers are already building algorithms that predict how you’ll metabolize the drug. One study showed these models could reduce adverse events by 30-40%. In five years, we might not just ask, “What generic are you on?” We might ask, “What’s your CYP3A4 genotype?”
Until then, awareness is your best tool. Know your numbers. Know your manufacturer. Know your body. Because with carbamazepine, the smallest change can have the biggest consequence.
Can I switch between different generic carbamazepine brands safely?
Switching between different generic carbamazepine brands carries risk, especially if you have epilepsy or a narrow therapeutic index. Studies show about 12% of patients experience therapeutic failure or new side effects after switching-even when the products meet FDA bioequivalence standards. This is because bioequivalence is tested in healthy adults on single doses, while real patients often have comorbidities, polypharmacy, or altered metabolism. If you’ve been stable on one generic, avoid switching unless monitored with blood tests. Always consult your neurologist before changing manufacturers.
Why does carbamazepine reduce the effectiveness of other drugs?
Carbamazepine strongly induces the CYP3A4 enzyme in the liver, which is responsible for breaking down about half of all medications. This includes anticoagulants like warfarin, immunosuppressants like cyclosporine, HIV drugs, birth control pills, and even some blood pressure medications. When carbamazepine boosts this enzyme, your body clears those other drugs faster, lowering their blood levels and reducing their effect. This induction starts within 48 hours and peaks in 2-3 weeks. Always review all medications with your doctor when starting or stopping carbamazepine.
How often should I get my carbamazepine level checked?
If you’re on carbamazepine, especially a generic version, get your blood level checked at baseline, then 7-14 days after any dose change or manufacturer switch. A second check at 4 weeks helps confirm stability. The therapeutic range is 4-12 mcg/mL, but individual needs vary. If your level changes by more than 15% after a switch, your dose may need adjustment-even if the milligram amount is the same. Patients with epilepsy, liver disease, or those on multiple medications should be monitored more frequently.
Are there genetic tests I should take before starting carbamazepine?
Yes. If you’re of Asian descent (particularly Han Chinese, Thai, Malaysian, or Filipino), you should be tested for the HLA-B*1502 gene before starting carbamazepine. Carriers have a 10-fold higher risk of developing Stevens-Johnson Syndrome, a life-threatening skin reaction. Screening is recommended by the FDA and is now standard practice in many clinics. Even if you’re not Asian, some genetic variants like CYP3A4*22 affect how you metabolize the drug, so pharmacogenetic testing may be useful if you’ve had unexpected side effects or poor response.
Can women on birth control take carbamazepine safely?
Carbamazepine reduces the effectiveness of hormonal birth control by increasing the breakdown of estrogen and progesterone. This raises the risk of unintended pregnancy. Women on carbamazepine should use non-hormonal contraception (like IUDs) or a backup method (like condoms). Even if you’ve been on the same birth control for years, carbamazepine’s enzyme induction can lower hormone levels over time. A 2021 study found women on carbamazepine had 22% higher rates of breakthrough seizures during certain phases of their cycle due to hormonal effects on metabolism. Always discuss contraception with your neurologist and OB-GYN.



17 Comments
Timothy Haroutunian
February 21 2026Let me tell you something about carbamazepine generics that no one wants to admit: the FDA’s bioequivalence standards are a joke. 80% to 125%? That’s a 45% window. Imagine if your insulin dose could vary by nearly half and still be considered 'the same.' You’d be dead. But somehow, with seizure meds, we just shrug and say 'it’s generic, it’s cheap.' I’ve seen patients go from stable to convulsing after a pharmacy switch. No one gets fired for cutting costs. But someone’s brain might.
And don’t get me started on how pharmacies rotate generics like it’s a game of musical chairs. I once had a patient on the same generic for three years-then suddenly, her pills looked different. No warning. No call from the doctor. Just a new bottle. She had a seizure two days later. The pharmacist said, 'It’s the same drug.' Yeah. And a hammer and a scalpel are both metal tools.
It’s not about money. It’s about control. The system is designed to push you toward the cheapest option, not the safest one. And if you’re poor, disabled, or uninsured? You’re just collateral. We talk about precision medicine like it’s some futuristic dream. Meanwhile, we’re letting people’s lives hinge on which factory made their pill this month.
I’ve been on carbamazepine for 18 years. I’ve switched generics five times. Each time, I had to relearn my body. Dizziness. Nausea. Brain fog. Then seizures. I keep a notebook. Manufacturer. Lot number. Date switched. Symptoms. Blood levels. I’ve become a pharmacist, a detective, and a trauma survivor all rolled into one. And I’m not special. I’m just the one who noticed.
The FDA doesn’t test for real-world conditions. No one tests how a 72-year-old with kidney disease and five other meds reacts to a new batch. No one tests how a woman on her period metabolizes it differently. No one tests how a guy with gastroparesis absorbs beads that are too big. They test healthy college kids on a single dose. That’s not science. That’s corporate negligence dressed up as regulation.
I’m not anti-generic. I’m pro-safety. If a drug has a narrow therapeutic window, it should be treated like a precision instrument. Not a commodity. But until the system changes, we’re all just rolling dice every time we refill a prescription.
Erin Pinheiro
February 23 2026okay so like i switched generics last month and i swear i felt like my brain was melting?? like i was so dizzy i couldnt even walk to the fridge and then i got this weird rash that looked like someone poured hot sauce on my skin?? and my dr just said 'oh its probably allergies' but i know better. i looked up the manufacturer and it was some company in india and now im convinced they used cheap fillers or maybe even fake drug?? like why would they risk our lives?? i even called the pharmacy and they said 'its all FDA approved' but like... is that even a real thing??
Michael FItzpatrick
February 24 2026What Timothy just said? That’s the raw truth. But let me add something deeper: carbamazepine isn’t just a drug-it’s a mirror. It reflects how broken our healthcare system is. We treat epilepsy like a math problem: dose = control. But it’s not. It’s a living, breathing, hormonal, genetic, metabolic dance. And we’re trying to choreograph it with a spreadsheet.
Women metabolize it faster. Men? Slower. Asians? Screen for HLA-B*1502. Elderly? Slower liver. Kidney disease? Slower clearance. Polypharmacy? Every other drug is a variable in the equation. And yet, we hand out the same pill, from the same bottle, with zero personalization.
Imagine if your car’s fuel efficiency changed every time you switched gas stations. You’d demand a mechanic. But with meds? We say 'take two.' No questions. No tests. No follow-up. It’s madness. And it’s not just dangerous-it’s disrespectful.
Therapeutic drug monitoring isn’t optional. It’s the bare minimum. If you’re on carbamazepine and you haven’t had a blood test since you started, you’re not being treated. You’re being experimented on.
And yes, some generics are fine. But you can’t know which ones unless you track them. Write down the manufacturer. Keep a log. Ask for DAW 1. Be your own advocate. Because no one else will.
We talk about personalized medicine like it’s science fiction. But here’s the reality: the science already exists. We just refuse to use it. That’s not incompetence. That’s choice.
Brandice Valentino
February 25 2026Ugh. Another one of these posts. 'Carbamazepine is dangerous.' Oh no, the system isn’t perfect. How dare you. Let me guess-you’re also one of those people who thinks the moon landing was fake and that fluoride is a government plot? Look, I’ve been on carbamazepine for 10 years. I switched generics twice. No issues. My neurologist says it’s fine. So why are we letting anecdotal horror stories from Reddit dictate policy? It’s not 1992. We have double-blind trials. We have pharmacokinetic data. The FDA doesn’t just let any junk through.
Also, why do people think 'brand name' is safer? The brand is just the original patent holder. The generics are identical. If they weren’t, they wouldn’t be approved. I get it-you’re scared. But fear doesn’t equal fact. Get a blood test if you’re paranoid. But don’t turn this into a conspiracy.
Larry Zerpa
February 27 2026Let’s cut through the noise. The entire narrative here is a manufactured crisis. The FDA approves generics based on rigorous bioequivalence standards. The 12% 'problem' rate? That’s within the margin of error for any chronic condition. Seizures happen. Side effects happen. People get stressed. They change laundry detergent. They sleep poorly. They skip meals. And suddenly it’s 'the generic changed.'
Real science? A 2020 meta-analysis of 17 studies found no significant difference in seizure control between generics and brand. Not one. Zero. Nada. The 'emergency room' statistic? It’s not even statistically significant when adjusted for confounders.
And let’s talk about HLA-B*1502. Yes, it’s real. But it’s rare. Outside of Southeast Asia, it’s under 0.5%. Yet we’re treating every patient like they’re a ticking bomb. This isn’t medicine. It’s fearmongering.
Pharmacists? They’re not out to kill you. They’re trying to keep costs down so people can afford their meds. If you want brand-name carbamazepine? Pay $800 a month. But if you’re on Medicaid? You get the generic. And that’s a good thing.
This post is a perfect example of how misinformation spreads. People read one story, panic, and turn it into a movement. Meanwhile, real patients who need access are being priced out by the very people screaming about 'safety.'
Stop weaponizing fear. Start trusting science.
Gwen Vincent
February 27 2026I just want to say thank you to everyone who shared their experiences. This isn’t just about a drug. It’s about being heard. I’ve been on carbamazepine for 7 years. I switched generics once. I had nausea and brain fog for 3 weeks. My doctor said it was 'probably stress.' I didn’t push back because I didn’t want to seem difficult.
But after reading this, I called my pharmacy. Asked for the manufacturer. Wrote it down. And next time I refill, I’m asking for DAW 1. I’m not trying to be a nuisance. I just want to stay stable.
If you’re reading this and you’re scared? You’re not alone. And you’re not overreacting. Your body knows when something’s off. Trust it. Advocate for yourself. Even if it feels awkward. Even if they roll their eyes. You deserve to be safe.
Nandini Wagh
March 1 2026Oh honey. You think this is bad? Wait till you hear about the time my cousin in Mumbai got switched to a generic made in a warehouse with no AC. The pills were sticky. The coating cracked. She had a seizure in the middle of a grocery store. They thought it was heatstroke. Took three days to figure out it was the meds.
Here’s the thing: the FDA doesn’t regulate overseas factories. They send inspectors once every 5 years. Meanwhile, 80% of our generic drugs come from China and India. You think they care about your brain? They care about profit margin.
And don’t even get me started on how pharmacies rotate generics to maximize profit. One week it’s Teva. Next week it’s Mylan. Next week it’s some company no one’s ever heard of. And they don’t tell you. Why? Because if you knew, you’d refuse.
This isn’t medicine. It’s a global lottery. And you’re the one holding the ticket.
Holley T
March 2 2026There’s a reason why carbamazepine is one of the few drugs that requires therapeutic drug monitoring. It’s not because it’s dangerous. It’s because it’s unpredictable. And the system refuses to acknowledge that.
Women on birth control? Their levels fluctuate with estrogen. Pregnant women? Clearance increases by 50%. Elderly? Reduced liver function. Kidney disease? Slower excretion. All of these factors are ignored in bioequivalence testing.
And yet, we treat all patients like they’re identical. That’s not medicine. That’s industrial manufacturing. You wouldn’t give the same insulin dose to a 90-pound teenager and a 250-pound man. Why do we do it with carbamazepine?
The FDA’s '80-125%' rule is a relic of 1980s policy. It was designed for antibiotics, not anticonvulsants. We need a new standard. One that accounts for therapeutic window, metabolic variability, and real-world physiology. Until then? Every switch is a gamble.
And yes-I’ve had a patient lose her job because she had a seizure after a pharmacy switch. Her insurance denied the claim because 'it was a generic.' I’m not exaggerating. This is happening every day.
Ashley Johnson
March 2 2026They’re lying to you. Carbamazepine isn’t just inducing enzymes. It’s being used to cover up something bigger. The pharmaceutical industry doesn’t want you to know that generics are often made with the same active ingredient but different binders-some of which are toxic. I’ve seen reports. They’re not public. But I have a friend who works at the FDA. He says they suppress data on liver damage linked to certain fillers.
And the HLA-B*1502 test? It’s not just for Asians. It’s for everyone. But they only screen certain groups because it’s cheaper. They know the real risk is higher. They just don’t want to pay for universal testing.
Also, the '12%' statistic? It’s a lie. It’s actually 37%. They just don’t report the severe cases. You think your seizures are 'coincidental'? Think again. The system is designed to keep you dependent. More seizures = more prescriptions. More prescriptions = more profit.
Don’t trust your doctor. Don’t trust the pharmacy. Don’t trust the FDA. They’re all part of the machine.
tia novialiswati
March 4 2026Hey, I just want to say-you’re not alone. 💛 I’ve been on carbamazepine since I was 16. I’ve switched generics 4 times. Each time, I felt off. I kept quiet because I didn’t want to be 'that patient.' But last year, I finally spoke up. I asked for my blood levels. They were at 3.8. Way below the range. I switched back to my old brand. No more seizures. No more dizziness.
Here’s what helped me: I made a little card. Manufacturer. Lot number. Date switched. Symptoms. I showed it to my neurologist. She said, 'Why didn’t you bring this sooner?'
You’re not overreacting. You’re being smart. Keep track. Ask questions. Tell your pharmacist. You’ve got this. I’m rooting for you. 🌻
Lillian Knezek
March 4 2026They’re watching you. Every time you refill, they track which generic you get. They’re building a profile. They know when you switch. They know when your levels drop. And then? They upsell you to a more expensive drug. Or they push you toward a 'new' treatment that’s actually just a repackaged version of the same thing.
I found out my pharmacy was selling my data to a drug company. They were paid to switch me to a specific generic. I didn’t even know. I thought it was random.
And the 'bioequivalence' studies? They’re funded by the manufacturers. Of course they say it’s safe. It’s like asking Big Tobacco if smoking causes cancer.
Don’t refill. Don’t trust. Don’t comply. Fight the system.
Maranda Najar
March 5 2026It is not merely a pharmacological phenomenon-it is a metaphysical betrayal of the patient-practitioner covenant. The commodification of life-saving medication has reduced the human body to a statistical abstraction, stripped of its sacred, idiosyncratic complexity. Carbamazepine, in its noble function as a stabilizer of neural chaos, has been perverted into a mere commodity, its therapeutic essence diluted by corporate indifference and regulatory apathy.
Each switch of a generic formulation is not merely a change in pill appearance-it is an existential rupture. The body, a temple of biochemical precision, is subjected to the capricious whims of supply-chain logistics and profit-driven substitution. To equate bioequivalence with therapeutic equivalence is not scientific-it is sacrilegious.
And yet, we are told to 'trust the system.' To bow before the altar of cost-efficiency. To accept the silent erosion of our neurological autonomy as the price of progress.
How many lives have been sacrificed on the altar of the 80–125% margin? How many seizures, how many ER visits, how many broken dreams, have been dismissed as 'anecdotal'?
The future of medicine must be rooted in reverence-for the body, for the individual, for the sanctity of therapeutic integrity. Until then, we are not patients. We are lab rats in a corporate experiment.
Christopher Brown
March 7 2026Stop whining. The FDA approves generics. If you can’t handle a pill switch, you shouldn’t be on a drug this complex. America’s healthcare system is the best in the world. If you want brand-name, pay for it. Stop blaming the system. Take responsibility.
Sanjaykumar Rabari
March 7 2026India makes good generics. You think America is better? Look at the opioid crisis. Look at the insulin prices. Your system is broken. We make safe drugs. You make money. That’s all.
Kenzie Goode
March 8 2026I just want to say-this whole thread made me cry. I’ve been on carbamazepine for 12 years. I’ve switched twice. Both times, I had no idea. I didn’t know to ask. I thought I was being 'good' by not complaining.
But last month, I had a seizure at work. I was terrified. I went to my neurologist. She asked if I’d switched generics. I didn’t even know what that meant.
She checked my levels. They were half of what they should be. We switched back. I’m stable now.
I’m not angry. I’m just… sad. That I didn’t know. That no one told me. That I had to almost die to learn.
If you’re reading this and you’re on carbamazepine? Please. Ask. Track. Write it down. Don’t wait until it’s too late.
Timothy Haroutunian
March 9 2026Kenzie’s comment hit me right in the chest. I didn’t have a seizure. But I lost my job. I was driving to work when I felt a weird aura-like static in my skull. I pulled over. Didn’t have a full seizure. But I had a panic attack. I called my boss. Said I was sick. I didn’t tell them it was my meds. I was ashamed.
They fired me two weeks later. 'You’re unreliable.' I couldn’t prove it was the pill. I didn’t have records.
Now I carry a printed sheet in my wallet. Manufacturer. Lot. Date switched. Blood level. Doctor’s contact. I don’t care if I look weird. I’d rather look weird than be dead.
And if you’re a doctor, pharmacist, or policymaker reading this? Stop treating patients like numbers. We’re not variables. We’re people. With jobs. Families. Lives.
Michael FItzpatrick
March 11 2026Timothy, your story is why I became a neurologist. I used to think the system was broken. Now I know it’s not broken-it’s designed this way. Profit before people. Efficiency before safety.
I’ve started a patient registry. I log every patient on carbamazepine. Their manufacturer. Their levels. Their switches. Their symptoms. I’ve collected 400+ cases. The pattern is undeniable. A drop in level >15% after a switch? 87% had clinical consequences.
We’re not asking for miracles. Just transparency. A label that says: 'This batch is from [Manufacturer]. Previous batch was [X]. Monitor levels.'
It’s not hard. It’s not expensive. It’s just human.