Generic vs Brand Name Drugs: A Guide to Labeling and Therapeutic Equivalence
Generic Drug Savings Estimator
Pro Tip Check the FDA Orange Book to ensure the generic has an "A" rating for therapeutic equivalence.
You've probably been at the pharmacy counter and had the pharmacist ask if you're okay with the generic version of your prescription. For some, it's a relief because it saves a significant amount of money. For others, it feels like a gamble. Is a cheaper pill really as effective as the one with the famous brand name on the box? The short answer is yes, but there are a few technical details about how they're made and labeled that explain why they look different even when they do the same thing.
The Basics of Therapeutic Equivalence
When we talk about therapeutic equivalence is the property of two or more drugs to produce the same clinical effect and pharmacological effect, we aren't just guessing. The FDA (U.S. Food and Drug Administration) has strict rules to ensure that a generic drug performs exactly like the brand-name version. To get approved, a generic must be the pharmaceutical equivalent, meaning it has the same active ingredient, strength, dosage form, and route of administration.
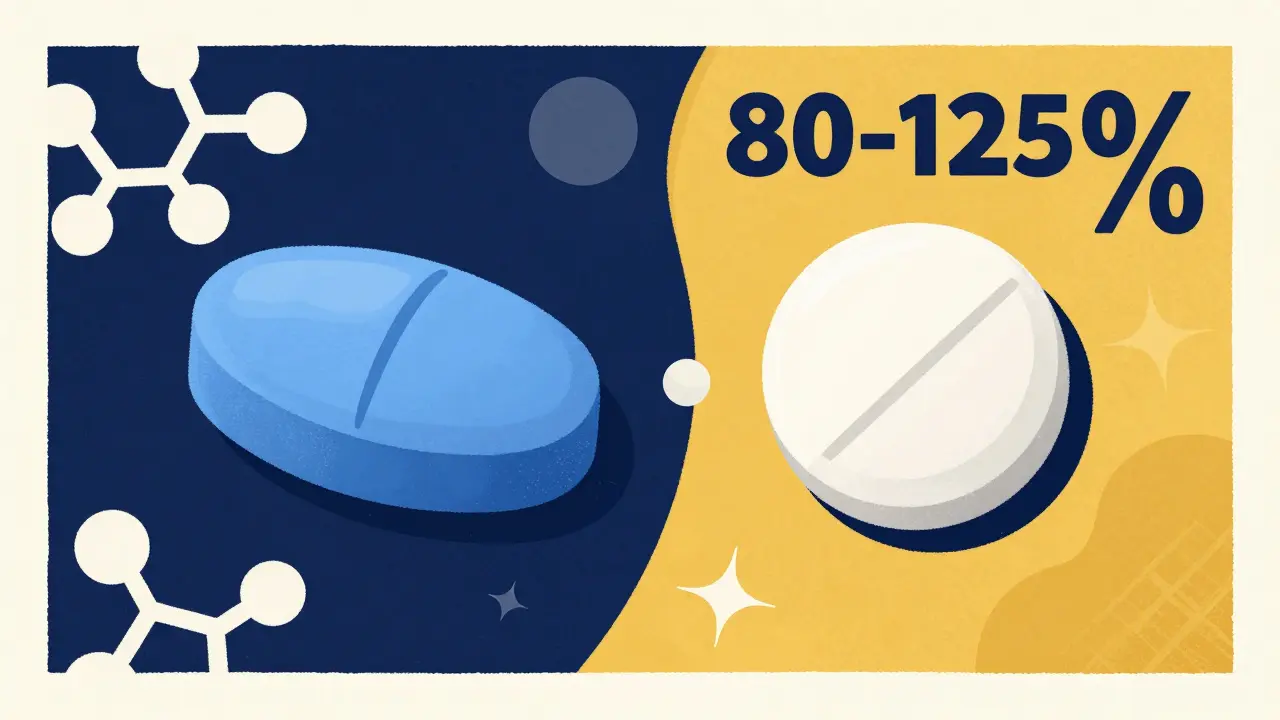
One of the most important hurdles for a generic drug is proving bioequivalence. This is a fancy way of saying the drug gets into your bloodstream at the same rate and to the same extent as the brand name. The FDA requires the pharmacokinetic profile-basically how the drug moves through your body-to fall within a tight window of 80-125% of the brand-name drug. Interestingly, some experts note that this 20% variability is actually tighter than the natural difference you might find between two different batches of the same brand-name drug.
Decoding the Label Differences
If you put a brand-name bottle and a generic bottle side-by-side, the first thing you'll notice is the name. Brand-name drugs use a proprietary name, like Prilosec, while the generic label lists the chemical name, such as omeprazole. This isn't a sign of lower quality; it's just how the law works. The brand company paid for the research and marketing to create a unique name, but once the patent expires, the chemical name becomes the standard.
Beyond the name, you'll see differences in the "inactive ingredients." While the active pharmaceutical ingredient (API) must be identical, the fillers, binders, and coloring agents (known as excipients) can vary. This is why a generic pill might be white and round while the brand is blue and oval. Trademark laws actually require generics to look different from the brand-name original. These changes in shape or color don't change how the medicine works in your system; they just prevent confusion in the marketplace.
| Feature | Brand-Name Drug | Generic Drug |
|---|---|---|
| Active Ingredient | Same | Same |
| Inactive Ingredients | Proprietary Mix | Varies by Manufacturer |
| FDA Approval Path | Full New Drug Application (NDA) | Abbreviated New Drug Application (ANDA) |
| Appearance | Trademarked Shape/Color | Must differ from Brand |
| Cost | High (Recovering R&D) | Low (Competitive Market) |
Why the Price Gap is So Huge
It can be shocking to see a brand-name drug cost hundreds of dollars while the generic costs a few bucks. For example, some data shows atorvastatin (the generic for Lipitor) costing as little as $4.00 at some retailers, while the brand version can run over $300 per month. Why the massive difference? It's not because the generic is "cheaply made."
Brand-name companies spend billions on clinical trials, research, and advertising. When they launch a drug, they get a patent that gives them a monopoly for a while so they can make that money back. Generic companies don't have to repeat those expensive clinical trials; they only have to prove that their version is bioequivalent to the original. Because multiple generic companies often compete for the same drug, the price drops drastically, making medication more accessible for everyone.
The Role of the Orange Book
If you're ever unsure if a generic is a true match, there is a gold-standard resource: the FDA Orange Book. Officially called "Approved Drug Products with Therapeutic Equivalence Evaluations," this database lists which generics are therapeutically equivalent to which brand-name drugs.
When looking at the Orange Book, you'll see ratings. An "A" rating means the generic is considered therapeutically equivalent to the brand. This means a pharmacist can typically swap them without needing a new prescription from your doctor, unless the doctor specifically wrote "Dispense as Written" (DAW) on the script.
When to Be Cautious: NTI Drugs
For 99% of medications, the switch is seamless. However, there's a small category called Narrow Therapeutic Index (NTI) drugs. These are medications where a tiny change in the blood concentration can be the difference between the drug working and it becoming toxic or failing completely.
Common examples include warfarin (a blood thinner), levothyroxine (for thyroid), and phenytoin (for seizures). Because these require such precision, some doctors prefer you stay on one specific manufacturer-whether it's the brand or a specific generic-to avoid any fluctuations. If you're taking an NTI drug and your pharmacy switches the generic manufacturer, it's a good idea to tell your doctor so they can monitor your levels more closely.

Complex Generics and Biosimilars
Not every drug is as simple as a pill in a bottle. Some medicines are complex molecules, like insulins or monoclonal antibodies. These are’t exactly "generics" because they are grown in living cells rather than mixed in a lab. These are called biosimilars. Because they are so complex, they are harder to replicate perfectly, and their approval process is different from a standard generic pill.
Other complex products, like the EpiPen, involve a delivery device. Even if the epinephrine inside is the same, the mechanism of the auto-injector must be exactly right to be approved. This is why you might notice fewer generic options for these high-tech delivery systems compared to standard tablets.
Are generic drugs less effective than brand-name drugs?
No. The FDA requires generic drugs to have the same active ingredient, strength, and bioequivalence as the brand-name version. Large-scale studies, including those published in JAMA Internal Medicine, have shown no significant difference in effectiveness for most therapeutic classes, including cardiovascular medications.
Why does my generic medication look different every few months?
This happens because your pharmacy may be switching between different generic manufacturers. Since different companies use different inactive ingredients (fillers and dyes) and are legally required to make their pills look different from the brand name, the color or shape may change even though the active medicine is the same.
Can I always switch to a generic?
In most cases, yes. However, for Narrow Therapeutic Index (NTI) drugs like levothyroxine or warfarin, you should consult your doctor first. Small variations in absorption between different manufacturers can affect these specific medications more than others.
What is the ANDA process?
The Abbreviated New Drug Application (ANDA) is the process generic drug companies use to get FDA approval. It is "abbreviated" because the company doesn't have to repeat the original clinical trials to prove the drug is safe and effective; they only have to prove it is bioequivalent to the already-approved brand-name drug.
How do I know if a generic is equivalent?
You can check the FDA's Orange Book. If a generic drug has an "A" rating, it is considered therapeutically equivalent to the brand-name product, meaning it can be substituted without compromising the therapeutic effect.
Next Steps for Patients
If you're looking to save money on your prescriptions, start by asking your doctor if a generic equivalent exists for your current medication. If you are taking a critical medication with a narrow window of effectiveness, ask your pharmacist to keep you on the same manufacturer to ensure consistency. For those managing multiple chronic conditions, checking the Orange Book or using a trusted pharmacy app can help you track which versions you're taking and help you spot any unexpected changes in pill appearance.